How do atoms link together? ⚛️
Welcome to Ask Professor Labcoat where Children’s Museum of Atlanta answers YOUR science questions!
Rebecca wants to know how do atoms link together?
Buckle up friends, because the answer to this question is really big for a subject so small.
We’re talking about atoms. These are the real building blocks of the universe. Atoms link to other atoms by sharing electrons, an EVEN SMALLER particle that atoms are made of! Atoms linking together create molecules. Molecules are one of the tiniest bits of a thing that can exist while still being the same thing. I know, that sounds crazy, but we’ll get there! The kind of molecules depends on what kind of atoms are bonded together, and how many of each there are.
There are 2 kinds of bonds atoms can make.
A covalent bond happens when atoms share an electron between them.
The other kind of bond is called ionic, and it happens when one atom gives an electron to another atom. These bonds are held together with electric forces.
Let’s go back to our crazy-sounding definition of a molecule: the tiniest bit of a thing that can exist while still being the same thing. Water is a molecule made of 2 hydrogen atoms and an oxygen atom. If you take away one of those atoms, you don’t have water anymore; you have something else. Likewise, adding atoms changes the molecule. If you add just one more oxygen atom to the water molecule, you now have a hydrogen peroxide molecule instead of water! That’s definitely not something you want to drink!
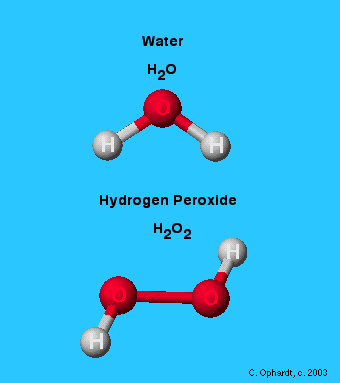
There are endless ways atoms and molecules can link together. These tiny bonds are what the entire universe is made of!
Thanks for your question! You can #AskProfessorLabcoat your science questions on Facebook, Twitter or Instagram!


